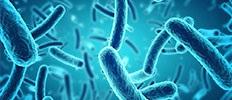
According to a new market research report, "Microbial Identification Market by Product & Service (Instrument & Software, Consumable, Service), Method (Phenotypic, Proteomic), Technology (Mass Spectrometry, PCR), Application (Diagnostic, Food Testing), End User (Hospital) – Global Forecast to 2025", The global microbial identification size is estimated to be USD 3.2 billion in 2020 and projected to reach USD 5.7 billion by 2025, at a CAGR of 12.2%.
Pathogenic bacteria alone cause about 350 million cases of foodborne diseases. About 48 million foodborne illnesses occur annually in the US alone, which leads to approximately 128,000 hospitalizations and 3,000 deaths every year. Poor water hygiene can cause 1.7 million deaths a year worldwide, and 9 out of 10 deaths occur in children. Most of these deaths, however, are observed in developing countries (NCBI).
According to the WHO, TB is one of the top 10 causes of death. A total of 1.5 million people died from TB in 2018 worldwide. Between 2000–2018, an estimated 58 million lives were saved through TB diagnosis and treatment.
Download PDF Brochure @ https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=10702182
Automated microbial identification systems are priced within the range of USD 50,000 to USD 75,000. Pharmaceutical companies require many such systems and, hence, the capital cost increases significantly.
Academic research laboratories generally cannot afford such systems as they have limited budgets. In addition, the maintenance costs and several other indirect expenses result in an overall increase in the total cost of ownership of these instruments. This hinders the mass adoption of automated microbial identification systems, especially by relatively small institutions.
[220 Pages Report] The global microbial identification market size is estimated to be USD 3.2 billion in 2020 and projected to reach USD 5.7 billion by 2025, at a CAGR of 12.2%. Growth in this market is largely driven by the rising incidence of infectious diseases and the increasing frequency of pandemics, technological advancements, increasing food safety concerns.
The current FDA approval process for instruments and consumables is very lengthy and complex. In May 2016, the Council of the European Union issued the new In Vitro Diagnostic Device Regulation (IVDR), which replaced the existing In Vitro Diagnostic Devices Directive (IVDD) 98/79/EC. IVDR was enforced in 2017 by the European council. The new rules will apply, starting May 2022, for IVDR. Companies will have five years to comply with the new regulations. These are more complex and stricter regulations.
Microbial identification is mainly used for human disease diagnosis. It delivers rapid and reliable results that help in the timely adoption of appropriate therapies. Microbial identification products can improve the management of infectious diseases, especially in areas with inadequate healthcare infrastructure. It is estimated that approximately 1,400 pathogens can cause human diseases.